The CCP1-CCP2 module pair of human C1r
C1r, the first protease of the classical pathway of the complement system, is a modular protein containing, among others, two CCP (complement control protein) modules and a serine protease (SP) domain at the C-terminus. We prepared the two CCP modules, CCP1 and CCP2, in their free form, as well as their tandem-linked construct, CCP1CCP2, to characterize their solution structure, conformational dynamics and cooperativity. The structures derived from NMR signal dispersion and secondary chemical shifts were in good agreement with those obtained by X-ray crystallography. The inter- and intramodular flexibility of the CCP modules proposed to play a central role in the autoactivation of this modular protease. Our NMR studies on the single CCP2 and the tandem CCP1-CCP2 constructs revealed that the CCP2 module has a well-folded structure in solution whereas the CCP1 module is much less-defined and more flexible. We showed that the CCP2 module stabilizes the CCP1 module through a well defined interface. NMR data suggest that in terms of folding, stability and dynamics, CCP1 is heavily dependent on the presence of its neighboring modules in intact C1r. Therefore, CCP1 could be a focal interaction point, capable of transmitting information towards its neighboring modules.
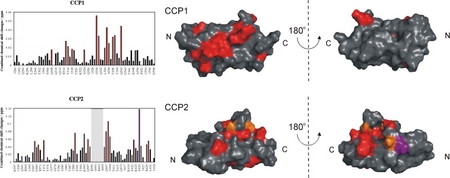
Interaction surfaces of CCP1 and CCP2 of human C1r
Cooperation with Prof. Dr. Péter Závodszky and Dr. Péter Gál, Structural Biophysics Group, Institute of Enzymology, Budapest
Kapcsolódó publikációk
-
András Láng , Balázs Major , Katalin Szilágyi , Zoltán Gáspári , Péter Gál , Péter Závodszky , András Perczel
Interaction between separated consecutive complement control modules of human C1r: implications for dimerization of the full-length protease.
FEBS Lett. 584(22):4565-4569. | DOI: 10.1016/j.febslet.2010.10.033 | PMID: 20970424 (2010) Kivonat -
András Láng , Katalin Szilágyi , Balázs Major , Péter Gál , Péter Závodszky , András Perczel
Intermodule cooperativity in the structure and dynamics of consecutive complement control modules in human C1r
FEBS J. 277(19):3986-3998. | DOI: 10.1111/j.1742-4658.2010.07790.x | PMID: 20796027 (2010) Kivonat