Skills
X-ray crystallography: a guide in structure based design of biologically active compounds
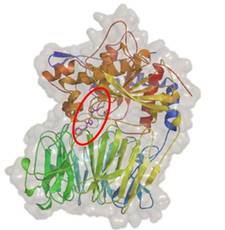
Crystallographic studies on prolyl-oligopeptidase (co-operation: Sanofi-Aventis)
It degrades proline-containing neuropeptides involved in the processes of memory and learning, it is thought to be involved in depression and control of blood-pressure.
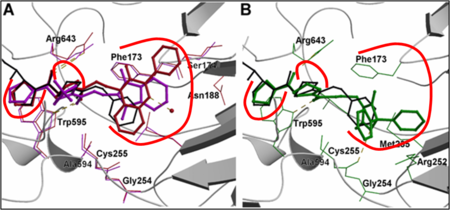
R-Pro-(decarboxy-Pro) type inhibitors, study on structure-activity relationships:
The optimal length of the linker chain of the R-group depends on the shape of the group in the terminal position.
Kánai K. et al. J. Med. Chem. (2008) 51: 7514-7522
Structure-function relationships by protein crystallography and molecular modelling
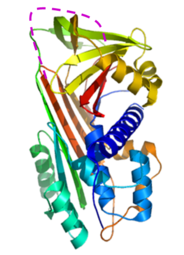
Crystallographic studies on C1-inhibitor (co-operation: HAS Institute of Enzymology)
A major downregulator of inflammatory processes in blood. Genetic deficiency of C1-inhibitor results in hereditary angioedema potentially lethal disease.
The crystal structure helped identifying the heparin binding site of C1 inhibitor.
Understanding the accelerating effect of heparin on the C1 inhibitor/protease reaction by a novel mechanism.
Beinrohr L. et al. J. Biol. Chem. (2007) 282: 21100 - 21109
Ligand binding of proteins at atomic level
Crystallographic studies on calmodulin-antagonist complexes (co-operation: HAS Institute of Enzymology)
The complexes formed with small molecules of different molecular frames show differences in the residues involved in binding. Most of the contacts are hydrophobic. In contrast to the other two compounds, KAR-2 is not a competitive antagonist, which is explained based on the structure.
Horváth I. et al. J. Biol. Chem. (2005) 280: 8266-8274
Harmat V. et al. J. Mol. Biol. (2000) 297: 747-755
Vértessy B.G. et al. Proteins: Struct. Funct. Gen. (1997) 28: 131-134

NMR-studies on dynamics, structure and stability of peptidic drug candidates
Penetratin have a possible role in the transport of drug molecules to the nucleus. Structures of the natural and mutant forms (NMR) revealed structure-function relationships.
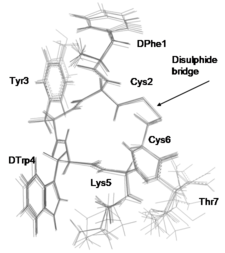
TT-232 is a somatostatin-analogue in clinical phase. Solving the its structure in water solution served better understanding the mechanism of its action.
Simon Á et al. Biochem. Biophys. Res. Comm. (2004) 316: 1059-1064
Letoha T et al. J. Mol. Recognit. (2003) 16: 272-279
Czajlik A et al. J. Pept. Sci. (2002) 8: 151-171
Cloning, expression, and crystallization of proteins
Preparation and purification of protein samples by molecular biology methods for NMR methods and X-ray crystallography
Isotope labelling
Expression of oligopeptides and miniproteins in fusion systems (fusion partner: ubiquitin or GFP)
Crystallization using vapour diffusion methods
High throughput crystallization using crystallization robot
![]()
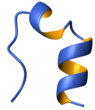
Tc5b, a 20 residue miniprotein was expressed in GFP fusion system for NMR spectroscopic studies



Crystals of a calmodulin complex, acylpeptide hydrolase, and MASP-2 grown in hanging drops
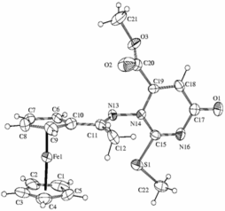
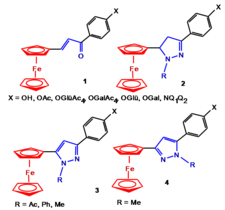
Synthesis of biologically active ferrocene derivatives
Ferrocenyl-substituated tiazolones, 1,3-tiazinones, pirimidones and imidazolones
Biologically active ferrocenyl-calcones and ferrocenyl-pirazoles. Some compounds exhibited remarkable antiproliferative effect against human leukemia cells (HL-60) in vitro.
Miklán Zs. et al. Biopolymers (Pept. Sci.) (2007) 88: 108-114
Zsoldos-Mády V. et al. ChemMedChem (2006) 1:1119-1125
Chemical crystallography: determining absolute configuration, studies on molecular recognition, structure-reactivity relationships
Metabolites of anti-parkinson drugs (co-operation: Sanofi-Aventis)
Aminopyridazinone regioisomers (co-operation: Semmelweis University, Budapest)
Structural basis of solid solution formation during chiral resolution (co-operation: Sanofi-Aventis)
Anwair M.A.S. et al. J. Agricult. Food. Chem. (2003) 51: 5262-5270
Barabás O. et al. Tetrahedron-Asymm.(2000) 11: 4061
Simon K. et al. Acta Cryst C54 (1998) 811-813
Main research topics:
- Structure, dynamics and interactions of proteins (crystallography, NMR and other spectroscopic methods)
- Structure and dynamics of protein building blocks (quantum chemical and spectroscopic methods)
- Structure based development of biologically active compounds
- Molecular modelling
- Biotechnology and chemical syntheses
- Chemical crystallography