NMR of synthetic polymers
We utilized NMR spectroscopy to study functional synthetic polymers, dendrimers, hyper branched and star polymers. Friedel–Crafts self‐grafting of polystyrene (PSt) under quasi-living carbocationic polymerization was utilized to develop a new rapid one‐pot method for the preparation of star‐shaped polyisobutylene (PIB). The formation of star polymers by this self‐grafting mechanism was proved by gel permeation chromatography equipped with online viscometer and 1H NMR spectroscopy. Unique, highly branched polyisobutylenes (PIB) were prepared via quasiliving carbocationic copolymerization of isobutylene and styrene (St) monomers. The resulting branched polymers were characterized and the branch formation was confirmed by gel permeation chromatography (GPC) and proton nuclear magnetic resonance spectroscopy (1H NMR). Telechelic polystyrenes (PSt) are useful intermediates for the synthesis of various macromolecular architectures. We reported the successful in situ synthesis of allyl-telechelic PSt via bulk quasiliving ATRP followed by direct allylation, not only in dichloromethane but also in a green solvent, benzotrifluoride. The quantitative transformations of the allyl terminals of PSt to hydroxyl and epoxy functionalities were described by using gel permeation chromatography and 1H-NMR spectroscopy.
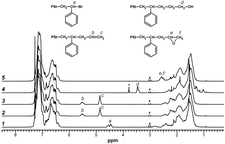
Structure and 1H-NMR spectra of telechelic polystyrenes
Cooperation with Dr. Béla Iván, Institute of Materials and Environmental Chemistry, Research Centre for Natural Sciences
Kapcsolódó publikációk
-
Gergely Kali , Márta Szesztay , Andrea Bodor , Béla Iván
A new synthetic method for the preparation of star-shaped polyisobutylene with hyperbranched polystyrene core
Macromol. Chem. Phys. 208: 1388-1393. (2007) Kivonat -
Gergely Kali , Márta Szesztay , Andrea Bodor , Béla Iván
Star and Hyperbranched Polyisobutylenes via Terminally Reactive Polyisobutylene‐Polystyrene Block Copolymers
Macromolecular Symposia 323(1), 37-41 | DOI: 10.1002/masy.201100116 (2013) Kivonat -
György Kasza , Györgyi Szarka , Andrea Bodor , Gergely Kali , Béla Iván
In Situ Terminal Functionalization of Polystyrene Obtained by Quasiliving ATRP and Subsequent Derivatizations
American Chemical Society 1285, 281-295 | DOI: 10.1021/bk-2018-1285.ch015 (2018) Kivonat