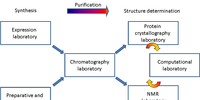
Expression laboratory
Gallery: Expression laboratory
Facility manager: Anna J. Kiss-Szemán, PhD
Members: István Bódy, Helga Tar-Pálfi, Márton Gadanecz, Dóra Nagy-Fazekas, Dr. András Láng, Dr. Nóra Taricska, Nóra Wagner, /en/members/former-members/107, /en/members/former-members/108
Modern gene and biotechnology made possible to expression (e.g. in bacterial systems), (re)design, point mutation, etc. of natural and denovo proteins. By using recombinant DNA techniques we are expressing short, 20-30 amino acid comprising miniproteins, as well as longer multidomain proteins composed of hundreds of residues. The nonselective isotope incorporation (15N and/or, 13C) as well as the residue specific isotope labeling is also possible. Miniproteins are expressed via fusion expression systems, with fusion partner proteins such as Ubiquitin, GFP, etc..
Laboratory News
-
-
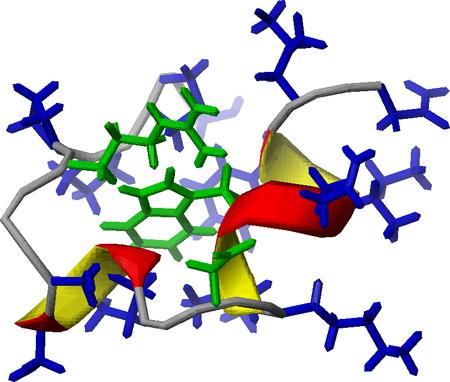
Great success of a small-, (mini)-protein
Expression and non-selective isotope labelling was a precondition of our research project aiming to characterise “invisible” intermediate structures of some Trp-cage miniproteins via temperature dependent NMR (1H-15N-HSQC) measurements.
Publications
-
Petra Rovó , Viktor Farkas , Orsolya Hegyi , Orsolya Szolomájer-Csikós , Gábor K. Tóth , András Perczel
Cooperativity network of Trp-cage miniproteins: probing salt-bridges
J Pept Sci 17(9):610-619. DOI: 10.1002/psc.1377 PMID: 21644245 (2011) Kivonat -
Péter Hudáky , Pál Stráner , Viktor Farkas , Györgyi Váradi , Gábor Tóth , András Perczel
Cooperation between a salt bridge and the hydrophobic core triggers fold stabilization in a Trp-cage miniprotein
Biochemistry 47(3):1007-1016. DOI: 10.1021/bi701371x PMID: 18161949 (2008) Kivonat -
Pál Stráner , Nóra Taricska , Mária Szabó , Gábor K. Tóth , András Perczel
Bacterial expression and/or solid phase peptide synthesis of 20-40 amino acid long polypeptides and miniproteins, the case study of Class B GPCR ligands
Curr. Prot. Pept. Sci. 17(2):147-155. DOI: 10.2174/1389203716666151102105215 | PMID: 26521952 (2016) Kivonat